Eutectic Composition
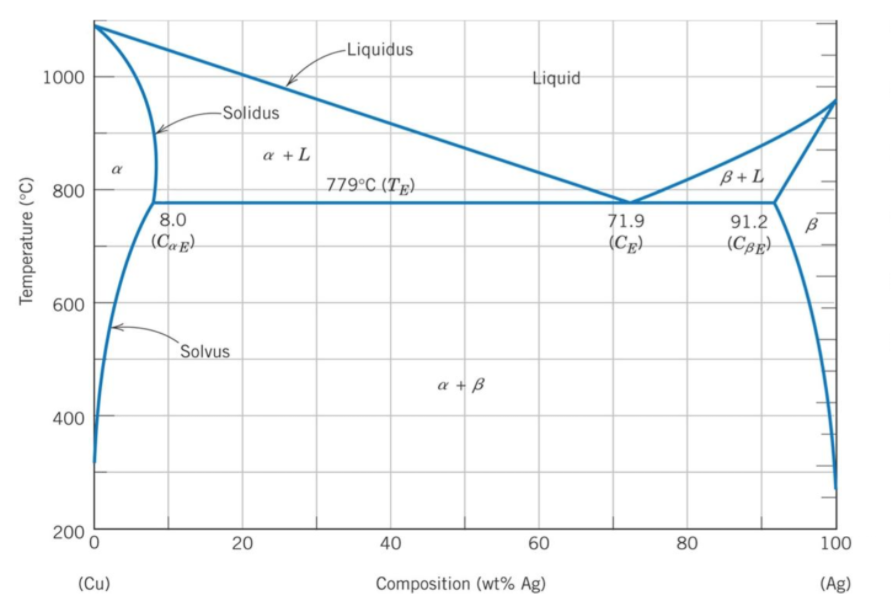
In the phase diagram below, what percentage composition of Ag-Cu will the alloy be in a eutectic composition?
Expand Hint
The eutectic composition is the point where an alloy will transform from a liquid phase directly into two solid phases.
Hint 2
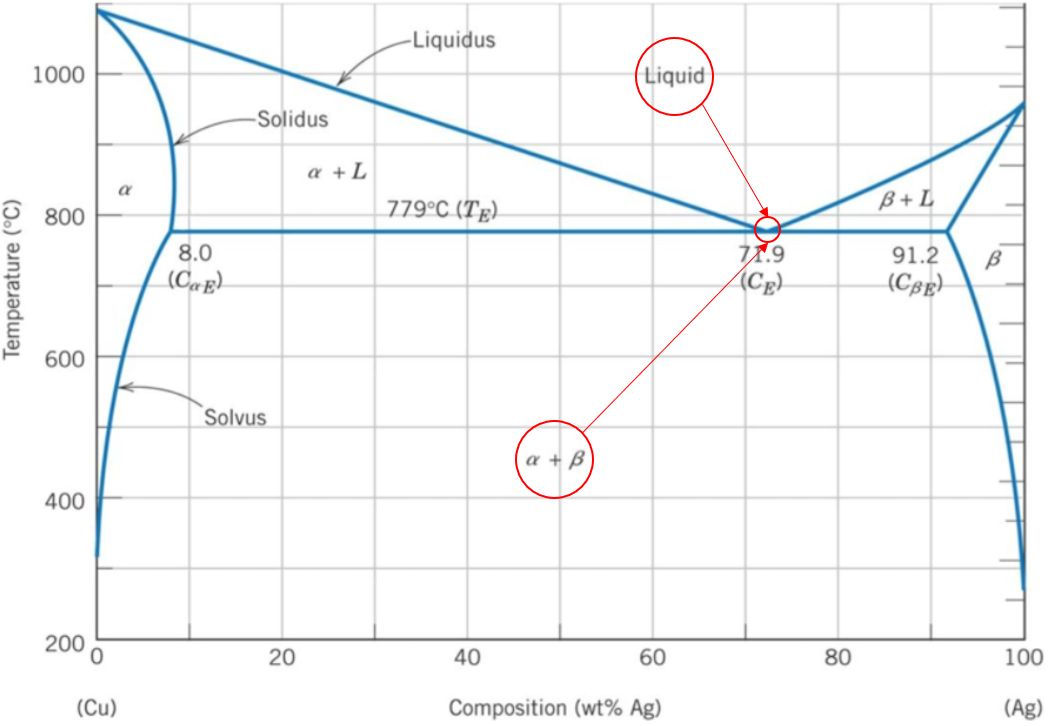
A binary phase diagram shows the phases (solid, liquid etc) formed by mixing two different elements over a range of temperatures. The eutectic composition is the point where an alloy will transform from a liquid phase directly into two solid phases. In other words, it is the lowest temperature point where the alloy will be completely melted. Based on the Ag-Cu phase diagram, the
$$Liquid\rightarrow \alpha + \beta$$
occurs at the 71.9% Ag (100%-71.9% Ag = 28.1% Cu) mark.
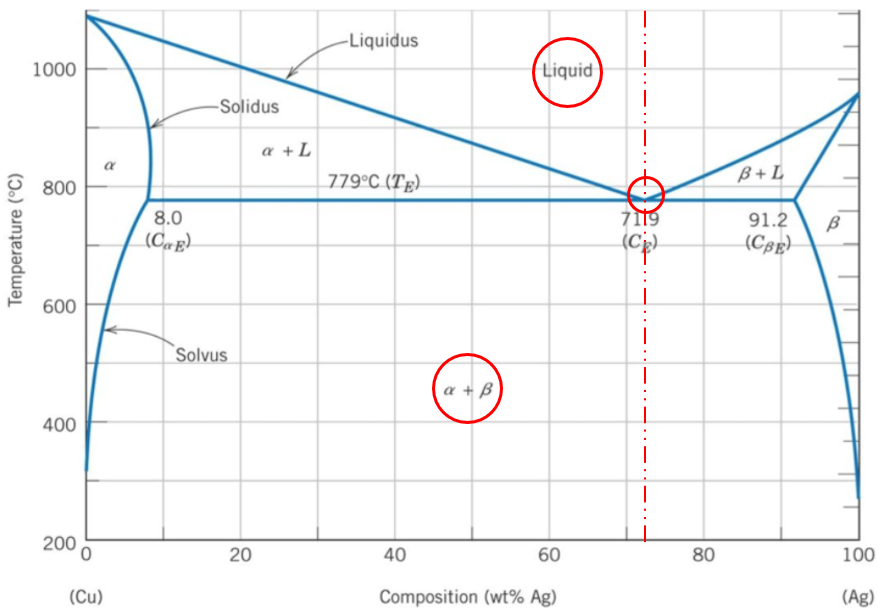
71.9 wt% Ag
Time Analysis
See how quickly you looked at the hint, solution, and answer. This is important for making sure you will finish the FE Exam in time.- Hint: Not clicked
- Solution: Not clicked
- Answer: Not clicked
Similar Problems from FE Section: Binary Phase Diagrams
232. Phase Diagram
372. Binary Phase Diagram